Periodic table chemistry lab 79/16/2023  
“Now that we have conclusively demonstrated the existence of element 113, we plan to look to the uncharted territory of element 119 and beyond, aiming to examine the chemical properties of the elements in the seventh and eighth rows of the periodic table, and someday to discover the island of stability,” he said. Kosuke Morita (pictured above), who led the Japanese group, has his sights set on that goal. 
Their huge nuclei would accelerate electrons close to the speed of light, almost certainly resulting in weird chemistry. WORKING WITH ELECTRICALLY POWERED LABORATORY EQUIPMENT. actinides belong in Periods 6 and 7, respectively, of the periodic table, between the elements of Groups 3 and 4. Stay connected with events during 2019’s International Year of the Periodic Table with information at the IYPT home page and the ACS IYPT page. Lead (atomic number 82) has a full outer shell, and so most elements heavier than it are unstable.īut heavier elements should include “islands of stability” where the outer shell of the nucleus is once again full. Prudent Practices in the Laboratory: Handling and Management of Chemical Hazards: Updated. Each of the more than 40 new elements, however, can be placed in a group of other elements with. Protons and neutrons are thought to occupy shells within the nucleus, and atoms are more likely to be stable if the outer shell is full. “The chemistry community is eager to see its most cherished table finally being completed down to the seventh row,” said Jan Reedijk, president of IUPAC’s inorganic chemistry division.Īpart from helping to flesh out the periodic table, the creation of superheavy elements puts theories of atomic structure to the test, and might one day yield stable new elements with strange properties. When we move from period 1 to period 7 in the periodic table. The discovery of the other three new elements has been credited to teams in Russia and the US, working collaboratively. The Periodic Table of Elements gives specific information chemical elements, their atomic. 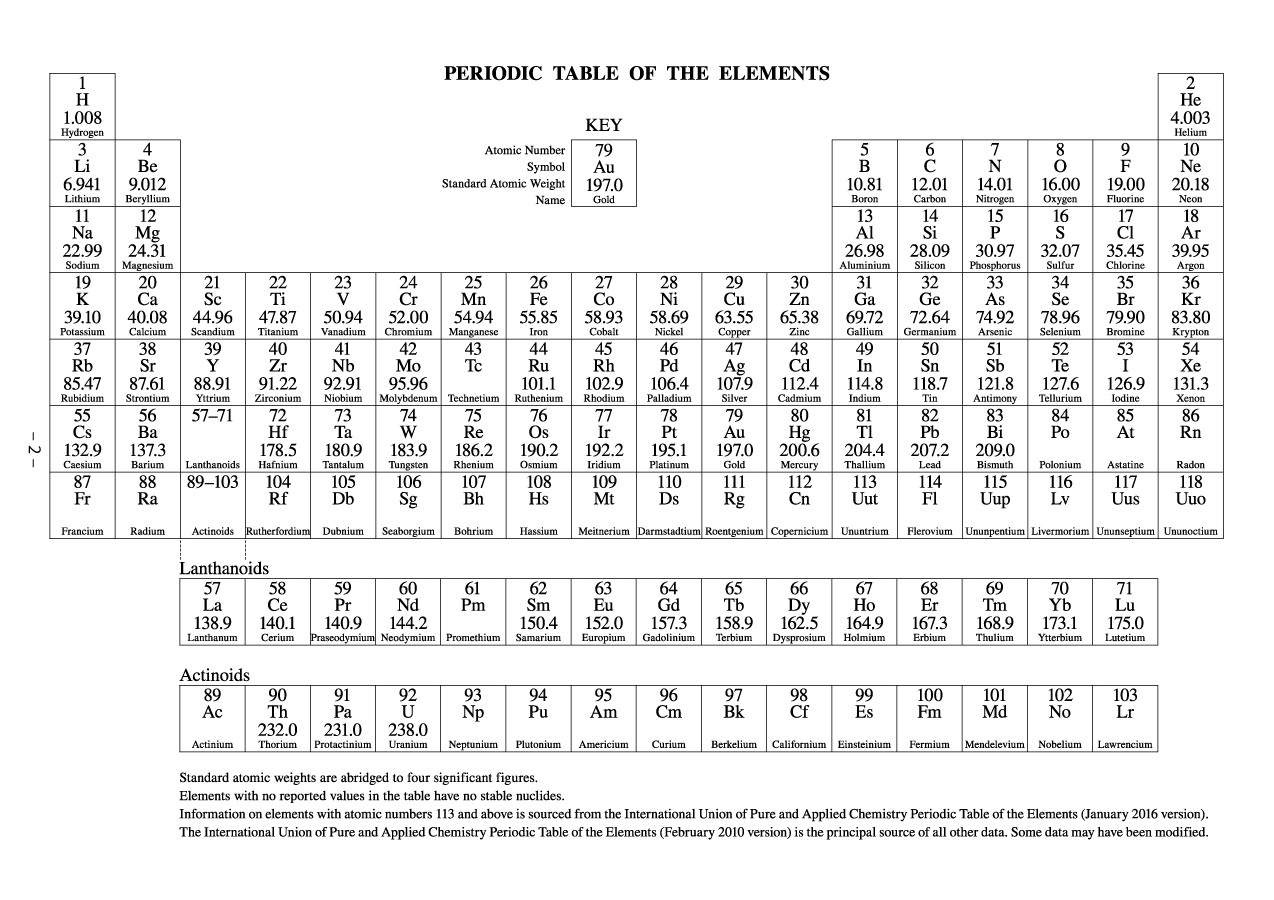
A team at the Joint Institute for Nuclear Research in Dubna, Russia, had claimed to have found it in 2003, but their result was ruled inconclusive. Controversyįor new elements to be accepted, there must be multiple detections in different experiments, and ruling on who made the discovery can be controversial.Ī collaboration at RIKEN in Wako, Japan, has been credited with finding element 113. The last time new elements were recognised was in 2011, when the periodic table gained flerovium (114) and livermorium (116). periodic table, in full periodic table of the elements, in chemistry, the organized array of all the chemical elements in order of increasing atomic numberi.e. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
